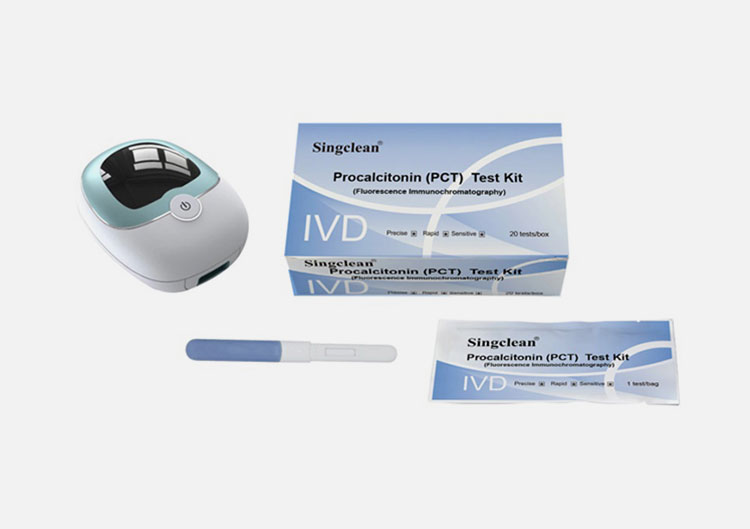
Procalcitonin (PCT) Test Kit
(Fluorescence Immunochromatography)
CE
[Product Name]
Procalcitonin (PCT) Test Kit (Fluorescence Immunochromatography)
[Packaging Specification]
10 tests/box, 20 tests/box, 25 tests/box, 30 tests/box, 50 tests/box, 100 tests/box.
[Intended Use]
The test kit is used for the quantitative determination of procalcitonin (PCT) in human serum, plasma or whole blood samples in vitro.
Procalcitonin (PCT) is the prohormone of calcitonin (CT), which is generally less than 0.1 ng/mL in the blood of healthy people, and its secretion increases after being stimulated by pro-inflammatory responses, especially after bacterial infection. Procalcitonin is an important marker that can specifically distinguish bacterial infection from inflammatory response caused by other reasons. Viral infection, allergy, autoimmune disease and transplant rejection do not cause significant elevation of procalcitonin, while local bacterial infection can result in moderate elevation of procalcitonin concentrations. In some cases (neonatal, multiple trauma, burns, major surgery, prolonged or severe cardiogenic shock), the elevation of procalcitonin may not be related to infection and usually returns to normal values quickly.
PCT is an ideal indicator for the auxiliary diagnosis of severe bacterial infection and septicopyemia, sepsis and other diseases, and has high sensitivity and specificity for systemic bacterial infection, septicopyemia, sepsis, etc.
[Test Principle]
This test kit uses fluorescence immunochromatography technology to quantitatively detect the content of procalcitonin (PCT) in serum, plasma or whole blood samples by double antibody sandwich method. After the sample is added to the test card, PCT in the sample reacts with the fluorescently labeled PCT monoclonal antibody 1 to form a complex, which continues forward chromatography along the nitrocellulose membrane to the test line, and forms a complex with the coated PCT monoclonal antibody 2 on the test line. Under the action of the excitation light source, the content of procalcitonin (PCT) in the sample is positively correlated with the fluorescent antibody signal, and the content of procalcitonin (PCT) in the sample is calculated by a fluorescence immunoanalyzer.
Principle of the supporting instrument: the measuring system of the instrument scans the binding area of the marker and the object to be tested on the detection card after the reaction to obtain an optical signal. Then the optical signal is measured and analyzed to quantitatively obtain the concentration of the measured substance.
[Main components]
No. | Name | Component |
1 | Procalcitonin (PCT) Test Kit (Fluorescence Immunochromatography) | Each bag contains a test card and a desiccant; the test card is composed of a t shell and a test strip, and the test strip consists of a sample pad, a fluorescent pad (fixed with fluorescently labeled PCT monoclonal antibody 1), nitrocellulose membrane (coated with PCT monoclonal antibody 2 and goat anti-mouse IgG), filter paper and PVC plastic plate |
2 | Whole blood diluent | 1 bottle of diluent for 10 tests/box, 20 tests/box, 25 tests/box, 30 tests/box; 2 bottles of diluent for 50 tests/box; 4 bottles of diluent for 100 tests/box; 3.0mL/bottle |
3 | ID card | 1pc/box. |
[Applicable instrument]
Fluorescence immunoanalyzer
[Storage conditions and shelf life]
The test kit should be stored in a sealed aluminum foil bag at 4℃~30℃, and the validity period is 18 months. After the foil bag is opened, the validity period is 1 hour.
[Sample requirements]
1. Human serum, plasma or whole blood samples; other body fluids and samples may not give accurate results.
2. Venous blood or fingertip blood should be collected under sterile conditions. It is recommended to use human serum or plasma for testing.
3. Anticoagulation with EDTA, sodium citrate or heparin is recommended for plasma and whole blood samples.
4. After the clinical blood samples are collected, the test must be completed within 4 hours at room temperature; serum and plasma can be stored at 2~8°C for 3 days and stored below -20°C for 5 months. Whole blood samples should not be frozen and stored at 2~8°C for 3 days. Avoid heat inactivating samples, and hemolyzed samples should be discarded.
5. Samples must be returned to room temperature before testing. Cryopreserved samples need to be completely thawed, rewarmed, and evenly mixed before use. Do not freeze and thaw repeatedly.
[Test method]
Please read the instructions for use of the test kit and the manual of the fluorescence immunoanalyzer carefully before use.
a) Bring the test kit and sample to be tested to room temperature.
b) Make sure the ID card matches the batch number of the kit, and insert the ID card into the card reading area of the instrument to read the information.
c) Open the inner package of the test card, take out the test card; draw 70μL of serum plasma sample, drop vertically to the test card sampling place, and start timing; draw 70μL whole blood, vertically drop it to the test card sampling place, and immediately add 1 drop whole blood diluent at the sample adding place and start timing.
d) After adding the sample, click "Start Test" on the screen of the fluorescence immunoanalyzer, and the test card will react at room temperature for 10 minutes; insert the test card into the test card slot of the fluorescence immunoanalyzer, and the instrument will automatically test the test card; The test results can be seen on the display screen of the immunoassay analyzer. Click "Print" on the screen to print the results.
[Positive judgment value or reference interval]
Reference value: <0.5ng/mL. The reference interval for plasma and whole blood samples is the same as that for serum samples. Due to differences in geography, race, gender and age, it is recommended that each laboratory establish its own positive judgment value or reference interval.
[Interpretation of test results] (for reference only, not used as clinical diagnostic criteria, test results need to be combined with other clinical and laboratory data for clinical diagnosis)
PCT mass concentration (ng/mL) | Clinical significance |
<0.05 | Normal value |
<0.5 | No or mild systemic inflammatory response. May be local inflammation or local infection. |
0.5-2 | Moderate systemic inflammatory response. Infection may be present. |
2-10 | Most likely sepsis, severe sepsis, or septic shock. There is a high risk of organ dysfunction. |
≥10 | Almost all severe bacterial sepsis or septic shock. Often accompanied by organ failure, there is a high risk of death. |
[Quality Control]
Quality control should be carried out on a regular basis to ensure the validity and accuracy of test results. The test kit does not contain quality control product, it is recommended to select appropriate quality control product for quality management.
[Limitations of the test method]
a) This kit is only for the test of human serum, plasma or whole blood samples. Abnormal hematocrit samples have an impact on the results of the whole blood test. The test results of serum, plasma and whole blood are not significant difference when the hematocrit is between 21% and 48%.
b) The test results need to be combined with other clinical and laboratory data, and if the PCT test results do not match the clinical assessment, further testing is required.
c) False positive results may be caused by: cross-reaction of antibody-like components in the blood; some nonspecific components in blood have similar antigenic determinants to capture fluorescent-labeled antibodies.
d) Hemoglobin, triglycerides and bilirubin in the blood sample can interfere with the test results, where the maximum allowable concentrations are 5g/L, 10g/L and 0.2g/L, respectively.
e) False negative results may be caused by: some unknown components shield the antigenic determinants from binding to the antibody; the unstable PCT antigen gradually degrades with time and temperature and cannot be recognized by the antibody. Valid test results depend on a good reagent and sample storage environment.
f) Other factors may also cause errors in test results, including technical reasons, operational errors and other sample factors.
[Product performance index]
a) Accuracy: The recovery rate is between 85% and 115%.
b) Linear range: within the linear range of 0.1ng/mL ~100ng/mL, the linear correlation coefficient r≥0.9900;
c) Blank limit: not higher than 0.05ng/mL;
d) Precision
In-batch precision: The coefficient of variation (CV) is not more than 15%;
Precision between batches: The relative range between batches is not more than 15%.
[Precautions]
1) This product is a single-use in vitro diagnostic reagent. Do not use expired products.
2) The ID card and the test card cannot be used together, if they are different batches.
3) Please read the instructions for use of the test kit and the manual of the fluorescence immunoanalyzer carefully before use.
4) The environmental temperature should be 15℃~30℃ when testing. The test card stored at low temperature needs to be returned to room temperature before opening to avoid moisture absorption.
5) The test card and fluorescence immunoanalyzer should avoid vibration and electromagnetic environment when using. It is normal for the instrument itself to vibrate during normal use.
6) There is a desiccant in the aluminum foil bag, do not take it orally.
7) Appropriate biosafety assurance procedures should be in place for those substances containing or suspected of containing infectious agents. The following are the relevant precautions: Handle the sample with gloves or disinfect this reagent; disinfect spilled samples or reagents with disinfectant; disinfect and handle all specimens, reagents and potential contaminants in accordance with local regulations.
8) This product is used for rapid clinical diagnosis and screening, and the final diagnosis should be made by a doctor after comprehensive examination indicators and clinical symptoms.
9)The used material must be discarded immediately after test. Disposal should be in accordance with accepted medical practice and applicable national, local or institutional guidelines.
10) Do not use if package is open or damaged.