[Product name]
MPH Absorbable Hemostatic Particles Applicator
[Models]
TQ-3
[Main structure and property]
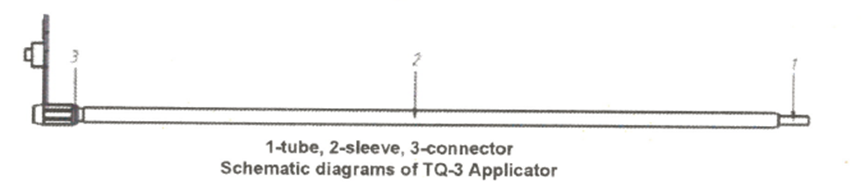
MPH Absorbable Hemostatic Particles Applicator (TQ-3) consists of a connector, a tube and a sleeve, and has a 25cm tube length and 23.5cm sleeve length. The applicator is made of medical polymer material, which has good biocompatibility, and is terminally sterilized by EO.
[Patient target groups]
The product is used in combination with MPH Absorbable Hemostatic Particles for patients requiring adjunctive hemostasis during surgery.
[Intended use]
The Applicator is intended to assist the delivery of MPH Absorbable Hemostatic Particles to the treatment site for endoscopic / laparoscopic surgical procedures.
[Direction for use]
To prevent product contamination prior to application, always follow aseptic techniques. For endoscopic / laparoscopic surgical procedures which spray MPH Absorbable Hemostatic Particles to control bleeding:
TQ-3 clinical application
1. Prior to initiating the procedure, visually inspect the package containing the Applicator for any signs of damage. If damage is visible, do not use the Applicator.
2. Remove the Applicator from the packaging.
3. Remove the MPH Absorbable Hemostatic Particles from its packaging, twist off its protective cap, and then install the nozzle.
4. Open the connector cap of the Applicator, then firmly press the MPH Absorbable Hemostatic Particles nozzle into the mouth of the Applicator connector. The Applicator is now ready for deployment.
5. Setup the trocar prior to use, position the Applicator tip at the site of bleeding, and begin delivering MPH Absorbable Hemostatic Particles by pumping the MPH bottle.
6. Remove the Applicator from the endoscope immediately after achieving the desired effect.
7. Avoid direct contact between the Applicator tip and blood at the surgical site to minimize the potential for clotting the tip during MPH Absorbable Hemostatic Particles application.
[Contraindication]
None reported to date.[Precautions]
1. This product is intended for single use only.
2. Do not use if the package is damaged.
3. Avoid the applicator tip contacting with tissue, organs, or blood.
4. The Applicator is sterilized by EO and should not be re-sterilized.
5. The used and expired products should be disposed of in compliance with pertinent government regulations regarding medical devices.
6. Please see the Instruction for use for MPH Absorbable Hemostatic Particles for complete information.
7. The user must be trained to obtain relevant qualifications before using.
8. In case of any serious device related incident, it shall be reported to the manufacturer or the competent authority.
[Storage]
This product should be stored in a dry and ventilated environment at a storage temperature of 2~30℃.
[Shelf Life]
It is valid for 24 months.
8.13.04.505-A0 Issue date: 2022-06-30